Countdown to the SI redefinition: How long would it take to eat a mole of Easter eggs?

In case you missed it, the redefinition of the International System of Units (SI) is going into effect next month on World Metrology Day, 20 May 2019. Each month we are bringing you a blog post featuring one of the units of the SI. This month we are focusing on a unit that has particular significance to chemists and therefore, our scientists at the UK National Measurement Laboratory at LGC: the mole, the unit used to measure the (usually very large) number of things, usually atoms or molecules.
602 214 076 000 000 000 000 000, or approximately 602 trillion, has 24 digits. As unimaginably large as this number may seem to us, we encounter trillions of things constantly in everyday life. In a regular sized balloon, for example, there are about 50 trillion air molecules (including about 10 trillion oxygen molecules). A standard painkiller contains around 1.7 trillion molecules of the active ingredient. And one litre of mineral water contains about 1 trillion sodium atoms.
As you can see, when we look at extremely small things such as atoms and molecules, we start having to deal with very large numbers. Physics and chemistry often deal with atoms and molecules but scientists don’t always want to have to juggle such large numbers. To make things more manageable, large particle numbers are combined into imaginary bundles for counting. This ‘bundle’ – the mole, the unit of an amount of substance – is our unit of the month for April.
Using this way of counting, instead of saying that a balloon contains “about 10 trillion oxygen molecules”, we can say there is “about 0.02 moles of oxygen". The conversion factor between the number of particles and the amount of substance is the so-called Avogadro constant, NA. This states that one mole contains 602 214 076 000 000 000 000 000 particles. Although we aren’t usually that interested in the exact number of oxygen atoms in a balloon, in other areas of life, such as medicines or food, it is critical that we know exactly how much of something there is and the mole becomes really important.
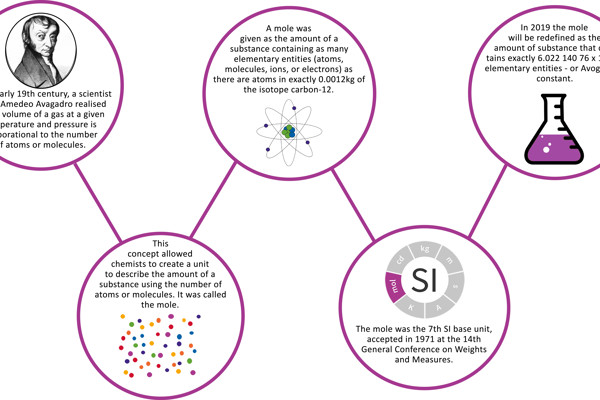
At the beginning of the 20th century, before the mole was introduced, terms such as "gram atom" and "gram molecule" were used to describe quantities of chemical elements or compounds. These quantities referred to the relative atomic or molecular weights and the reference value for these relative weights was the atomic weight of oxygen, generally agreed to be 16. However, there was some disagreement between the physics and chemistry communities as to which exact definition of oxygen was meant [1]. In 1960 the International Union of Pure and Applied Physics (IUPAP) and the International Union of Pure and Applied Chemistry (IUPAC) agreed to assign the value of 12 to the so-called atomic weight of the isotope [2] of carbon with the mass number 12 (12C), correctly referred to as the relative atomic mass Ar(12C). From then on, this was used as the reference value.
At the suggestion of these international bodies responsible for agreement on standards and atomic weights (IUPAP, IUPAC and ISO), a definition of the mole as a unit for the quantity of the substance was developed at the end of the 1960s. The definition was adopted into the International System of Units (SI) by the General Conference on Weights and Measures (CGPM) in 1971. It reads:
- The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon 12; its symbol is "mol".
- When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles, or specified groups of such particles.
From 20 May 2019, the definition of the mole will be as follows:
The mole, symbol mol, is the SI unit of amount of substance. One mole contains exactly 6.022 140 76 × 1023 elementary entities. This number is the fixed numerical value of the Avogadro constant, NA, when expressed in the unit mol−1 and is called the Avogadro number.
The amount of substance, symbol n, of a system is a measure of the number of specified elementary entities. An elementary entity may be an atom, a molecule, an ion, an electron, any other particle or specified group of particles.
So what does this change really mean for the mole? Even though the two definitions look very different at first glance, in reality the answer is: not much. As for all the other SI base units, a constant (the Avogadro constant) is the basis of the definition from World Metrology Day (May 20th) onwards. Yet even if the Avogadro constant is not explicitly mentioned in the current definition, it is already closely linked, as the Avogadro constant corresponds to the number of particles contained in 12 g of the carbon isotope 12C.
To get an idea of what a mole represents, we’ve performed an Easter-themed thought experiment.
If we collected 602 trillion molecules of water (or one mole of water), we would be able to fit a whole mole into a single egg cup. But if you placed just 0.01 mole of eggs upright next to each other, you could cover an area as large as the surface of our sun [3]. In fact, with a whole mole of eggs you could stack 100 layers of densely packed eggs on the sun. Which would not only be difficult to do but would end up with a huge amount of scrambled eggs!
But how many eggs is that? Put it this way, even if every person alive today found an Easter egg every minute it would take over a hundred million years to find one mole of Easter eggs.
Can you think of any more number games and thought experiments to describe the mole? Get in touch on Twitter (@NML_ChemBioGC) to tell us your ideas. Whoever sends the 602-trillionth message gets a hand-painted Easter egg!
[1] Oxygen occurs in nature in three different isotopes [2]. By means of physical measuring methods these isotopes can be separated due to their different masses. In physics, the value 16 was attributed to the lightest oxygen isotope. In chemistry, the value 16 was attributed to the natural mixture of isotopes, which for them was the naturally occurring element oxygen.
[2] Isotopes of an element have the same number of protons in the atomic nucleus, but different numbers of neutrons. The atoms of different isotopes of an element therefore have different weights. Oxygen has three different natural isotopes; all have 8 protons in the atomic nucleus, the isotope 16O has 8 neutrons, 17O has 9 neutrons and 18O has 10 neutrons in the atomic nucleus in addition to the 8 protons.
